A Rare Reaction: When an Inhaler Triggers Sweet Syndrome
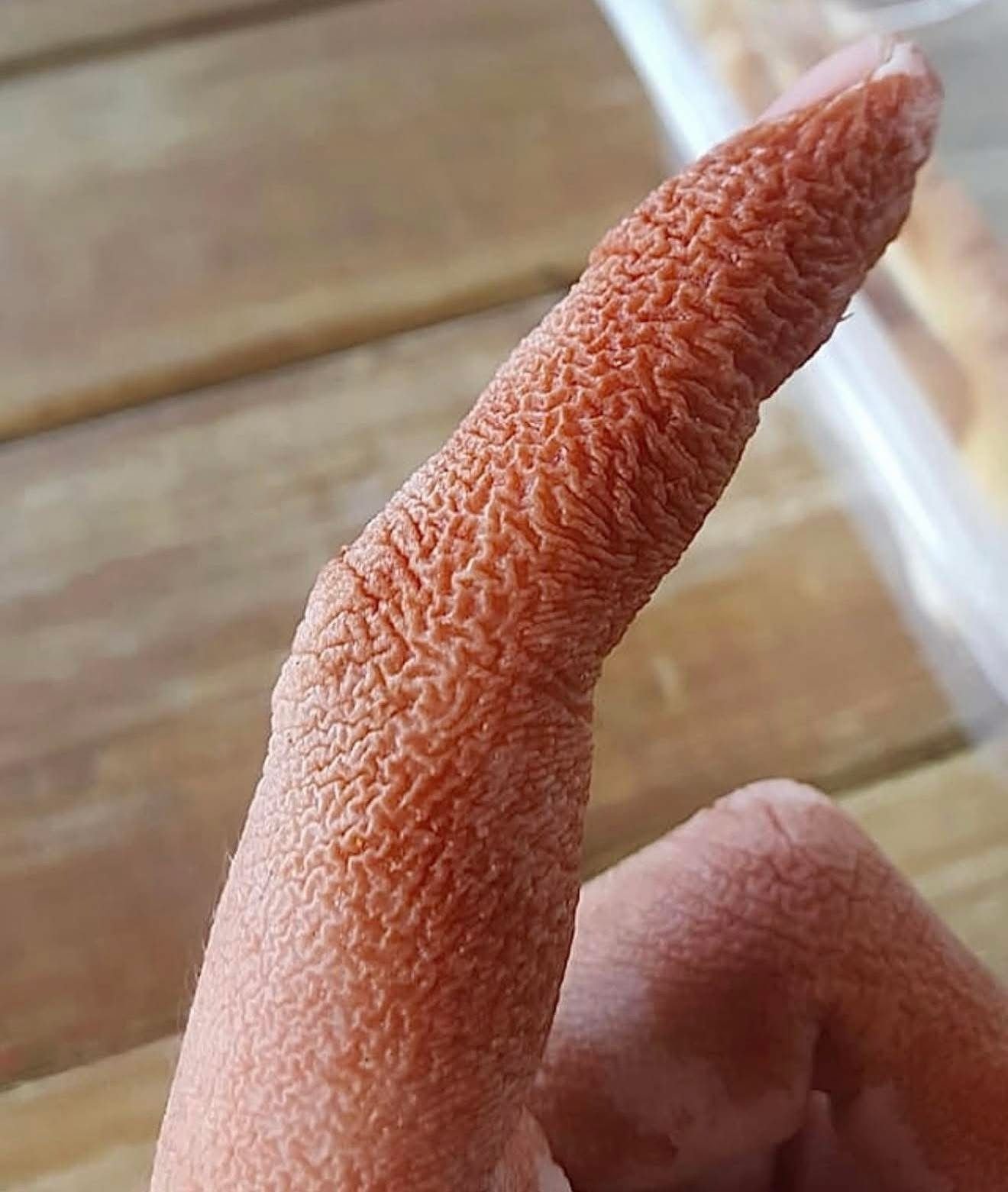
What started as a routine adjustment in a patient’s COPD inhaler rapidly unfolded into a puzzling medical mystery. Within just two days, a 55-year-old woman developed intensely painful, bright red patches across her face and neck, accompanied by a mild fever.
While the visible symptoms subsided quickly, the biopsy results revealed an unexpected diagnosis—Sweet syndrome, a rare immune-driven skin disorder.
This unusual case may mark the first time an inhaled medication has been linked to this condition, raising new questions about hidden risks in commonly prescribed treatments.
The patient, with a medical history including hypertension and COPD, had been stable on enalapril and formoterol inhaler for years. Due to declining lung function, her pulmonologist switched her inhaler to a combination of indacaterol and glycopyrronium. Just 48 hours after starting the new inhaler, she presented with painful erythematous plaques on her face and neck, plus a low-grade fever. She denied new cosmetic use, diet changes, or cold symptoms but mentioned recent sun exposure with proper protection.
Referred urgently to dermatology, she discontinued the inhaler. Blood tests ruled out infections and autoimmune markers, while a skin biopsy confirmed Sweet syndrome. Oral corticosteroids were prescribed, leading to rapid symptom improvement within two days.
Also known as acute febrile neutrophilic dermatosis, Sweet syndrome is characterized by sudden onset of painful, red papules or plaques—often asymmetrical—primarily affecting the face, neck, upper torso, and hands. It typically presents with systemic symptoms such as fever and elevated white blood cells.
While its exact cause remains unclear, Sweet syndrome is believed to stem from cytokine-driven immune responses, triggered by infections, cancers, or certain medications—including antibiotics, antiepileptics, and vaccines. Treatment with corticosteroids usually brings swift relief.
This case stands out because inhaled therapies have never before been documented as a trigger. Differential diagnoses like contact dermatitis, lupus, or urticaria were carefully excluded.
Sweet syndrome’s rarity and its potential link to serious systemic diseases mean it’s often overlooked in initial diagnoses. This case highlights the critical role primary care providers play in identifying unusual reactions tied to medication changes, ensuring timely referrals and treatment.
This case serves as an important reminder that even medications we commonly consider routine and generally safe—such as inhalers used for asthma and other respiratory conditions—can, in rare instances, trigger unexpected and potentially serious immune-mediated reactions. While these treatments are widely prescribed and have significantly improved the quality of life for millions of patients worldwide, they are not entirely free from risk. On very rare occasions, individuals may develop unusual inflammatory responses that can affect the skin, the bloodstream, and other organ systems, sometimes in ways that are difficult to predict or immediately recognize.
One such rare but clinically significant condition is Sweet syndrome, also known as acute febrile neutrophilic dermatosis. This disorder is characterized by the sudden onset of painful skin lesions, often accompanied by fever and an elevated white blood cell count, particularly neutrophils. The condition can appear abruptly and may initially be mistaken for infections, allergic reactions, or other dermatological disorders. Because of its variable presentation, Sweet syndrome can present a diagnostic challenge, especially when it is triggered by medications that are not commonly associated with severe immune responses.
In the context of medication-induced Sweet syndrome, inhalers and other respiratory therapies are not typically considered high-risk agents. However, the immune system is highly complex and can respond unpredictably in certain individuals due to genetic predisposition, underlying health conditions, or concurrent medication use. When such reactions occur, they often require careful clinical evaluation, including a detailed patient history, laboratory testing, and sometimes a skin biopsy to confirm the diagnosis. Recognizing the potential link between recent medication changes and the sudden appearance of symptoms is crucial for timely and accurate diagnosis.
Clinicians play a vital role in identifying these rare but important adverse drug reactions. A high index of suspicion is essential, particularly when patients present with acute skin eruptions, fever, or systemic symptoms shortly after starting or adjusting a medication regimen. In many cases, early recognition can significantly influence patient outcomes. Prompt discontinuation of the suspected triggering medication, combined with appropriate medical treatment such as corticosteroids, can lead to rapid improvement and full recovery.
This case also highlights the importance of maintaining a broad differential diagnosis when evaluating dermatological symptoms. While common conditions are often considered first, healthcare providers must also remain open to less frequent possibilities, especially when standard treatments fail to produce expected results. Expanding diagnostic awareness to include conditions like Sweet syndrome ensures that patients receive more accurate diagnoses and more effective treatment plans in a timely manner.
From a pharmacovigilance perspective, cases like this underscore the importance of continuous monitoring and reporting of adverse drug reactions. Even medications with well-established safety profiles can occasionally produce rare side effects that only become apparent through accumulated clinical experience and reporting systems. By documenting and analyzing such cases, the medical community can improve its understanding of drug safety and enhance future patient care.
For patients, these rare reactions can be alarming, particularly because they often develop suddenly and without warning. Clear communication between healthcare providers and patients is therefore essential. Patients should be encouraged to report any new or unusual symptoms promptly, especially after starting a new medication or changing a dosage. Early reporting can make a significant difference in diagnosis and treatment outcomes.
In addition, this case emphasizes the importance of individualized medicine. Every patient responds differently to treatment, and what is safe for one person may not be entirely suitable for another. Factors such as immune system variability, underlying medical conditions, and environmental influences all play a role in how medications are tolerated. Understanding this variability helps clinicians approach treatment with greater caution and adaptability.
Research into drug-induced Sweet syndrome remains ongoing, with the aim of better identifying risk factors and underlying mechanisms. Scientists continue to explore how certain medications may trigger abnormal immune responses in susceptible individuals. Although such cases are rare, they provide valuable insights into the complex interactions between drugs and the immune system.
Ultimately, this case reinforces a fundamental principle of clinical medicine: vigilance saves lives. Even well-established and commonly used medications should be approached with awareness of their rare but possible adverse effects. By remaining attentive to sudden changes in a patient’s condition, particularly skin-related symptoms following medication adjustments, healthcare professionals can ensure early detection, accurate diagnosis, and effective intervention.
In conclusion, recognizing atypical causes of Sweet syndrome expands diagnostic awareness and enhances clinical practice. It allows healthcare providers to move beyond common assumptions and consider a wider range of possibilities when evaluating patient symptoms. This broader perspective not only improves diagnostic accuracy but can also lead to faster, life-changing interventions that significantly improve patient outcomes and safety.


Leave a Reply